Synthesis of Ce–Ni/sepiolite with excellent adsorption and catalytic oxidation performance toward dyes
Abstract
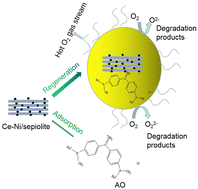
A novel and highly efficient wastewater treatment process for the removal of industrial dyes was proposed in this study. The method involved the regeneration of Ce–Ni/sepiolite, which acted as an adsorbent/catalyst, exhausted with the cationic dye auramine O (AO)—a frequently-used industrial dye—under a stream of O2 hot gas. The Ce–Ni/sepiolite adsorbent/catalyst was successfully synthesized by impregnation and was characterized by XRD, SEM, XPS, and BET techniques. An orthogonal experimental design method was applied to optimize the synthesis conditions of Ce–Ni/sepiolite. These synthesis conditions used the regeneration efficiency/rate of the catalyst as the evaluation index under heated O2 gas stream regeneration. Ce–Ni/sepiolite exhibited remarkable adsorption and catalytic oxidation degradation qualities for AO. The regeneration rate of as-prepared Ce–Ni/sepiolite reached 112.9% after 1 hour at 500 °C under 6 L min−1 of hot gas. The batch adsorption capacities were found to be 210.60 mg g−1 (25 °C), 243.90 mg g−1 (35 °C) and 263.16 mg g−1 (45 °C). 45 °C seemed to allow for the optimum adsorption capacity. Ce–Ni/sepiolite also showed high dynamic adsorption capacity, able to treat 48 bed volume (BV) of the dye solution with a residual concentration of 0 mg L−1 when the height to diameter ratio is 2.5. The adsorption and regeneration mechanisms were proposed. The as-synthesized Ce–Ni/sepiolite can be used as an efficient adsorbent and catalyst in wastewater treatments. This further supports the prospective usability of the sepiolite adsorbent for dye removal in future industrial applications.


 Please wait while we load your content...
Please wait while we load your content...