Dendritic hollow nitrogen-doped carbon nanospheres for oxygen reduction at primary zinc–air batteries†
Abstract
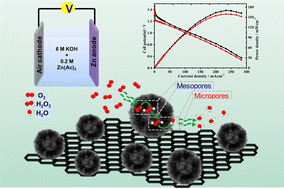
The cost and scarcity of platinum-based electrocatalysts are the major drawbacks for the large-scale commercialization of low-temperature fuel cells. Transition metal or metal-free heteroatom-doped carbon catalysts are promising alternative electrocatalysts for the oxygen reduction reaction in alkaline fuel cells. Dendritic hollow N-doped carbon spheres with a large inner accessible surface area are synthesized from the polyindole precursor on a dendritic fibrous nano-silica template. The thus-synthesized metal-free N-doped carbon material exhibits the highest onset (∼1.0 V vs. RHE) and half-wave potential (0.84 V vs. RHE), surpassing the ORR activity of the state-of-the-art Pt/C catalysts. The pore size plays an important role in the activity and in controlling the reaction pathway, wherein mesopores catalyze the partial reduction of O2 to H2O2, and micropores facilitate the H2O2 reduction to H2O. A zinc–air battery using the synthesised catalyst as the ORR catalyst exhibits the highest power density of 175 mW cm−2 with excellent specific capacity (951 mA h gzn−1 @ 20 mA cm−2), depending on the surfactant used and synthesis method. The ORR mechanism on the dendritic hollow N-doped carbon, based on the synthesis method and surfactants, is discussed.


 Please wait while we load your content...
Please wait while we load your content...