Nitroxide radical surfactants enable electrocatalytic oxidation of fatty alcohols in water†
Abstract
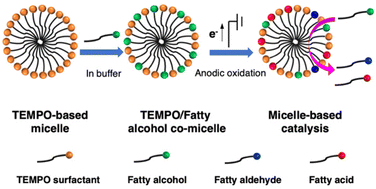
Oxidation of fatty alcohols is an important reaction to produce industry-needed compounds such as fatty aldehydes and fatty acids. While electrochemical oxidation of alcohols in water represents an environmentally friendly approach, it is a great challenge for the oxidation of long-chain fatty alcohols because of their low reactivity and water-solubility. Here, a typical nitroxide radical, 2,2,6,6-tetramethylpiperidyl-1-oxy (i.e., TEMPO), functionalised surfactants have been developed and used as efficient catalysts for the electrochemical oxidation of long-chain fatty alcohols to fatty aldehydes and fatty acids. The surfactants with varying aliphatic chain-length (i.e., C4-NO˙ to C14-NO˙) increase the partition of fatty alcohols in water through the formation of co-micelles. This process enables the TEMPO-mediated electrocatalytic oxidation of fatty alcohols in water with an increased turnover frequency (TOF) and a 102–103 fold increase in the apparent reaction rate kobs compared with non-surfactant TEMPO derivative 4-acetamido-TEMPO (ACT). Bulk oxidation showed the electrocatalytic reaction cascade of fatty alcohols to corresponding fatty aldehydes and eventually to fatty acids, with conversions as high as 93%.
- This article is part of the themed collection: 2023 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...