Self-flooding behaviors on the fuel cell catalyst surface: an in situ mechanism investigation†
Abstract
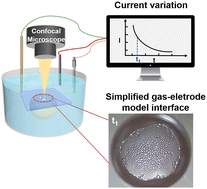
Water management in fuel cells (FCs) has been a long-standing bottleneck for device performance and durability. To get flooding pictures at the catalyst layer, a model reaction system was established for the in situ observation of the gas/electrode interface, by using a pre-pinned reactant gas bubble on an underwater transparent Pt/ITO electrode via confocal microscopy, during the oxygen reduction reaction (ORR)/hydrogen oxidation reaction (HOR). The flooding phenomenon, in terms of the formation of micrometer-sized droplets at the bubble/electrode interface, was observed during the alkaline ORR and acidic HOR as ion-generation reactions, while the non-flooding phenomenon (no obvious droplets) was observed for the alkaline HOR and acidic ORR as ion-consumption reactions. The unexpected flooding phenomenon was attributed to the decrease in saturated vapor pressure driven by the generation of ions (H+ for acidic HOR and OH− for alkaline ORR), leading to the condensation of the water vapor at the bubble/electrode interfaces; however, the non-flooding phenomenon was due to the elevated vapor pressure caused by the consumption of ions at the interfaces. For all the flooding cases, the real-time current was proportional to the uncovered gas/electrode interface area, and a higher aerophilicity of the electrode would lead to a higher current density. The present investigation offers a novel perspective for water management in FCs.


 Please wait while we load your content...
Please wait while we load your content...