Homoleptic complexes of isocyano- and diisocyanobiazulenes with a 12-electron, ligand-based redox capacity†
Abstract
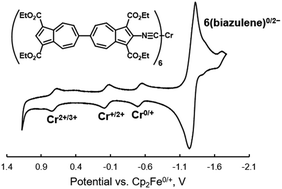
Oligo- and polyazulenes are attractive π-conjugated building blocks in designing advanced functional materials. Herein, we demonstrate that anchoring one or both isocyanide termini of the redox non-innocent 2,2′-diisocyano-6,6′-biazulenic π-linker (1) to the redox-active [Cr(CO)5] moiety provided a convenient intramolecular redox reference for unambiguously establishing that the 6,6′-biazulenic scaffold undergoes a reversible one-step 2e− reduction governed by reduction potential compression/inversion. Treatment of bis(η6-naphthalene)chromium(0) with six equiv. of 2-isocyano-1,1′,3,3′-tetraethoxycarbonyl-6,6′-biazulene (6) or [(OC)5Cr(η1-2,2′-diisocyano-1,1′,3,3′-tetraethoxycarbonyl-6,6′-biazulene)] (11) afforded homoleptic Cr(0) complexes 13 and 14 with a 12e− (per molecule) ligand-based reduction capacity at mild E1/2 of −1.29 V and −1.15 V vs. Cp2Fe0/+, respectively. The overall reversible redox capacity varies from 15e− for the mononuclear complex 13 to 21e− for the heptanuclear complex 14. The latter “nanocomplex” has a diameter of ca. 5 nm and features seven Cr(0) centers interlinked with six 2,2′-diisocyano-6,6′-biazulenic bridges. The X-ray structure of [(OC)5Cr(2-isocyano-1,1′,3,3′-tetraethoxycarbonyl-6,6′-biazulene)] (7) indicated a 43.5° interplanar angle between the two azulenic moieties. Self-assembly of 11 on a Au(111) substrate afforded an organometallic monolayer film of 11 featuring approximately upright orientation of the 2,2′-diisocyano-6,6′-biazulenic linkers, as evidenced by ellipsometric measurements and the RAIR signature of the C4v-symmetric [(–NC)Cr(CO)5] infrared reporter within 11. Remarkably, comparing the FTIR spectrum of 11 in solution with the RAIR spectrum of 11 adsorbed on Au(111) suggested electronic coupling at a ca. 2 nm distance between the Cr(0) and Au atoms linked by the 2,2′-diisocyano-6,6′-biazulene bridge.


 Please wait while we load your content...
Please wait while we load your content...