Dynamical characterization and multiple unbinding paths of two PreQ1 ligands in one pocket†
Abstract
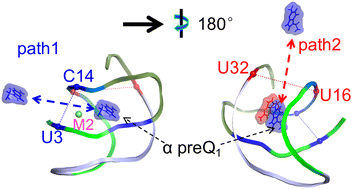
Riboswitches naturally regulate gene expression in bacteria by binding to specific small molecules. Class 1 preQ1 riboswitch aptamer is an important model not only for RNA folding but also as a target for designing small molecule antibiotics due to its well-known minimal aptamer domain. Here, we ran a total of 62.4 μs conventional and enhanced-sampling molecular dynamics (MD) simulations to characterize the determinants underlying the binding of the preQ1-II riboswitch aptamer to two preQ1 ligands in one binding pocket. Decomposition of binding free energy suggested that preQ1 ligands at α and β sites interact with four nucleotides (G5, C17, C18, and A30) and two nucleotides (A12 and C31), respectively. Mg2+ ions play a crucial role in both stabilizing the binding pocket and facilitating ligand binding. The flexible preQ1 ligand at the β site leads to the top of the binding pocket loosening and thus pre-organizes the riboswitch for ligand entry. Enhanced sampling simulations further revealed that the preQ1 ligand at the α site unbinds through two orthogonal pathways, which are dependent on whether or not a β site preQ1 ligand is present. One of the two preQ1 ligands has been identified in the binding pocket, which will aid to identify the second preQ1 Ligand. Our work provides new information for designing robust ligands.


 Please wait while we load your content...
Please wait while we load your content...