Five novel supramolecular assemblies constructed from the thiazolothiazole extended viologen moiety†
Abstract
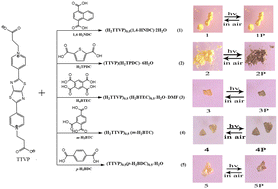
Lately, an increasing number of research groups have been inspired by the construction and optical capabilities of 2,5-bis (4-pyridine)-thiazolo [5,4-d] thiazole. Herein, five novel supramolecular assemblies based on the extended viologen moiety (TTVP, 2,5-bis (pyridinium-4-yl) thiazolo [5,4-d] thiazole propionate) and different benzenecarboxylic acids (1,4-H2NDC = 1,4-naphthalenedicarboxylic acid; H2TPDC = thiophene-2,5-dicarboxylic acid; H4BTEC = 1,2,4,5-benzenetricarboxylic acid; m-H3BTC = 1,2,4-benzenetricarboxylic acid; p-H2BDC = 1,4-benzenedicarboxylic acid) are composited by a self-assembly reaction, namely (H2TTVP)0.5(1,4-HNDC)·2H2O (1), (TTVP)(H2TPDC)·6H2O (2), (H2TTVP)0.5(H2BTEC)0.5·H2O·DMF (3), (H2TTVP)0.5(m-H2BTC) (4), (TTVP)0.5(p-H2BDC)0.5·H2O (5). The electron-deficient extended viologen moiety and the electron-donating benzenecarboxylate moieties are hydrogen-bonded to build the D–A system. Although five supramolecular assemblies comprise the same TTVP moiety, only 2–5 exhibit reversible photochromic behavior on account of the formation of extended viologen radicals via electron transfer (ET). Meanwhile, the photo-controlled fluorescence and the inkless and erasable printing capabilities are discussed.


 Please wait while we load your content...
Please wait while we load your content...