Chromium measurement in pharmaceutical samples: a comparative study of three new membrane electrodes with different electron-ion exchangers
Abstract
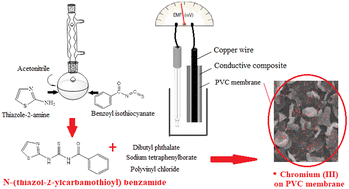
The present study deals with synthesis of N-(thiazol-2-ylcarbamothioyl) benzamide. It was utilized as a neutral ionophore for designing three types of chromium(III) sensors including coated wire ion selective electrodes (CW-ISEs), ion selective electrodes with liquid internal electrolyte (LIE-ISEs), and solid-state ion selective electrodes (SS-ISEs). UV-visible spectrophotometry was used to confirm the affinity of N-(thiazol-2-ylcarbamothioyl) benzamide to chromium(III). It was found that a membrane with a composition of 2% NaTPB, 8% ionophore, 60% DBP, and 30% PVC showed the best performance and a Nernstian slope of 21.6 mV per decade. Scanning electron microscopy was used to assess the PVC membrane morphology. The existence of chromium(III) in the liquid membrane matrix was proved by energy-dispersive X-ray spectroscopy. Detection limits for SS-ISE (1 × 10−6 M) and CW-ISE (1 × 10−6 M) were enhanced relative to LIE-ISE (1 × 10−5 M). All three electrodes showed a response time of about 5 s. The sensors' applicable pH range was 4.0–6.0. Fourier transform infrared spectra recorded through the electrode membrane showed that chromium(III) ion can interact with sulfur, nitrogen and oxygen atoms of N-(thiazol-2-ylcarbamothioyl) benzamide. The sensors were utilized as indicator electrodes in chromium(III) potentiometric titration with ethylenediaminetetraacetic acid and for directly measuring chromium(III) in some pharmaceutical samples.


 Please wait while we load your content...
Please wait while we load your content...