Highly stable and isomorphic donor–acceptor stacking in a family of n-type organic semiconductors of BTBT–TCNQ derivatives†
Abstract
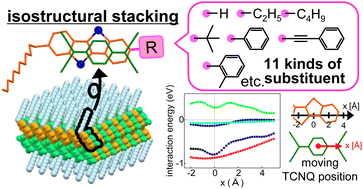
Herein, we present the common structural features of a family of semiconducting molecular donor–acceptor (DA) compounds based on alkylated fused-ring thienoacenes. Crystal structure analyses were conducted for 22 DA compounds of variously substituted benzothieno[3,2-b][1]benzothiophenes (BTBTs), unsubstituted dithieno[3,2-b:2′,3′-d]thiophene, and unsubstituted benzo[1,2-b:4,5-b′]dithiophene as donors, which were combined with 7,7,8,8-tetracyanoquinodimethane (TCNQ) and its fluorinated derivatives (FmTCNQ, m = 0, 1, 2, and 4) as acceptors. Fourteen DA compounds of (substituted BTBT)(TCNQ derivative) formed isomorphous layered molecular packing, where the intermolecular stacking arrangements between the planar skeletons of BTBT and TCNQ were common to each other, and the various substituents played supplementary roles. Density functional theory calculations were conducted to investigate the intermolecular forces between π-electron cores along the DA stacks. Dispersion interactions were the dominant intermolecular attraction, showing weak stacking position dependence, while the short-range orbit–orbit interaction was always repulsive, irrespective of stacking. An important finding is that the orbit–orbit interaction has a relatively strong position dependence and leads to common structural features in the DA compounds. We discuss the origin of the isostructural nature of molecular DA compounds, which is crucial in searching for and exploring unique combinations of molecules with superior semiconducting characteristics.


 Please wait while we load your content...
Please wait while we load your content...