Solar driven efficient direct conversion of methane to multicarbon oxygenates†
Abstract
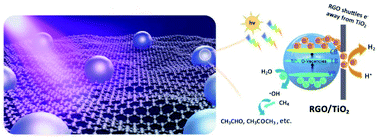
Utilizing sunlight to produce valuable chemicals from methane serves as a promising green strategy, exploiting hydroxyl radicals generated by water photooxidation to accomplish methane C–H activation. However, the selective and efficient synthesis of multicarbon compounds for direct methane conversion remains highly challenging. Herein, we present a composite catalyst composed of reduced graphene oxide/titanium dioxide (RGO/TiO2) with abundant oxygen vacancies for photocatalytic methane conversion to C2 and C3 oxygenates using only water vapor as an oxidant under mild conditions. The optimized catalyst achieves aldehyde and acetone yields of 289.6 and 191.2 μmol gcat−1 h−1, respectively, and multicarbon oxygenate selectivity as high as 97% under simulated solar irradiation, far superior to those of state-of-the-art photocatalytic processes. Comprehensive characterization demonstrates the synergistic effect between the π–π-conjugated substrate of RGO and the oxygen vacancies of TiO2, expanding visible light absorption and increasing charge separation/transfer efficiency, resulting in outstanding photocatalytic activity. Furthermore, density functional theory calculations suggest favorable chain growth routes through methane–methanol–CO-oxygenate pathways. Our findings provide insights into the design of highly active and stable photocatalysts for selectively converting methane into desirable value-added chemicals under ambient conditions.


 Please wait while we load your content...
Please wait while we load your content...