Abstract
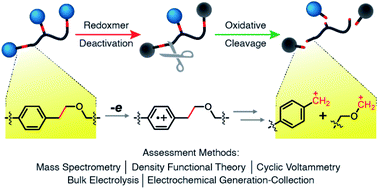
Irreparable chemical damage to redox-active monomers, oligomers, and polymers (i.e. redoxmers) limits the lifetime of energy storage devices (e.g. redox flow batteries) by causing electrode and membrane fouling, as well as irreversible capacity loss. To predictably restore device and materials performance, it is desirable to add programmed destruction capabilities into these damage-prone materials. Here, we report the use of triggerable retrograde reactions to achieve programmable end-of-life function in redoxmers. Retrograde reactions are intended to break up damaged redoxmer materials into more soluble small-molecule constituents to prevent irreparably fouling battery interfaces. We investigated the redox-triggered mesolytic cleavage of homobenzylic ethers (HBEs) for this purpose. Combining experimental methods with simulations, we probed the influence of ring substituents on the programmable bond-scission behavior of HBEs. Variation of para-substituents on the HBEs allowed us to modify the oxidation potential, degradation pathway, and electrochemical mechanisms of the generated products. Given its ideal oxidation potential, we selected the para-methoxy-based HBE as the cleavage scaffold and integrated it with dialkoxybenzene, TEMPO, viologen, and para-nitrobenzene redox centers for compatibility, bulk deconstruction, and electrode defouling tests. Upon applying a high oxidation potential, the redox-active pendant is cleaved from the backbone via HBE bonds; of the four redox centers tested, three of them remained fully redox active. Viologen-appended redox active polymers with HBE linkers were cleaved to fully separate the redox centers from the backbone. Polymer-filmed electrodes were partially restored following HBE oxidation defouling. These studies highlight the co-design of new materials functions for developing sustainable energy storage materials.
- This article is part of the themed collection: In Memoriam of Susan A. Odom (16 November 1980 – 18 April 2021)


 Please wait while we load your content...
Please wait while we load your content...