Backbone-controlled LUMO energy induces intramolecular C–H activation in ortho-bis-9-borafluorene-substituted phenyl and o-carboranyl compounds leading to novel 9,10-diboraanthracene derivatives†
Abstract
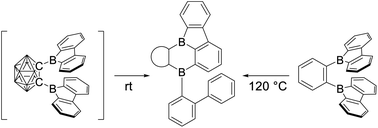
The choice of backbone linker for two ortho-bis-(9-borafluorene)s has a great influence on the LUMO located at the boron centers and, therefore, the reactivity of the respective compounds. Herein, we report the room temperature rearrangement of 1,2-bis-(9-borafluorenyl)-ortho-carborane, C2B10H10-1,2-[B(C12H8)]2 ([2a]) featuring o-carborane as the inorganic three-dimensional backbone and the synthesis of 1,2-bis-(9-borafluorenyl)benzene, C6H4-1,2-[B(C12H8)]2 (2b), its phenylene analog. DFT calculations on the transition state for the rearrangement support an intramolecular C–H bond activation process via an SEAr-like mechanism in [2a], and predicted that the same rearrangement would take place in 2b, but at elevated temperatures, which indeed proved to be the case. The rearrangement gives access to 3a and 3b as dibora-benzo[a]fluoroanthene isomers, a form of diboron polycyclic aromatic hydrocarbon (PAH) that had yet to be explored. The isolated compounds 2b, 3a, and 3b were fully characterized by NMR, HRMS, cyclic voltammetry (CV), single-crystal X-ray diffraction analysis, and photophysical measurements, supported by DFT and TD-DFT calculations.


 Please wait while we load your content...
Please wait while we load your content...