Nitrogen activation and cleavage by a multimetallic uranium complex†
Abstract
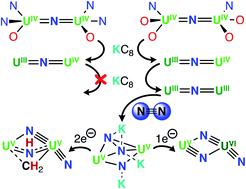
Multimetallic-multielectron cooperativity plays a key role in the metal-mediated cleavage of N2 to nitrides (N3−). In particular, low-valent uranium complexes coupled with strong alkali metal reducing agents can lead to N2 cleavage, but often, it is ambiguous how many electrons are transferred from the uranium centers to cleave N2. Herein, we designed new dinuclear uranium nitride complexes presenting a combination of electronically diverse ancillary ligands to promote the multielectron transformation of N2. Two heteroleptic diuranium nitride complexes, [K{UIV(OSi(OtBu)3)(N(SiMe3)2)2}2(μ-N)] (1) and [Cs{UIV(OSi(OtBu)3)2(N(SiMe3)2)}2(μ-N)] (3-Cs), containing different combinations of OSi(OtBu)3 and N(SiMe3)2 ancillary ligands, were synthesized. We found that both complexes could be reduced to their U(III)/U(IV) analogues, and the complex, [K2{UIV/III(OSi(OtBu)3)2(N(SiMe3)2)}2(μ-N)] (6-K), could be further reduced to a putative U(III)/U(III) species that is capable of promoting the 4e− reduction of N2, yielding the N24−complex [K3{UV(OSi(OtBu)3)2(N(SiMe3)2)}2(μ-N)(μ-η2:η2-N2)], 7. Parallel N2 reduction pathways were also identified, leading to the isolation of N2 cleavage products, [K3{UVI(OSi(OtBu)3)2(N(SiMe3)2)(![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N)}(μ-N)2{UV(OSi(OtBu)3)2(N(SiMe3)2)}]2, 8, and [K4{(OSi(OtBu)3)2UV)(
N)}(μ-N)2{UV(OSi(OtBu)3)2(N(SiMe3)2)}]2, 8, and [K4{(OSi(OtBu)3)2UV)(![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N)}(μ-NH)(μ-κ2:C,N-CH2SiMe2NSiMe3)-{UV(OSi(OtBu)3)2][K(N(SiMe3)2]2, 9. These complexes provide the first example of N2 cleavage to nitride by a uranium complex in the absence of reducing alkali metals.
N)}(μ-NH)(μ-κ2:C,N-CH2SiMe2NSiMe3)-{UV(OSi(OtBu)3)2][K(N(SiMe3)2]2, 9. These complexes provide the first example of N2 cleavage to nitride by a uranium complex in the absence of reducing alkali metals.
- This article is part of the themed collection: Most popular 2022 main group, inorganic and organometallic chemistry articles


 Please wait while we load your content...
Please wait while we load your content...