NickelII-catalyzed asymmetric photoenolization/Mannich reaction of (2-alkylphenyl) ketones†
Abstract
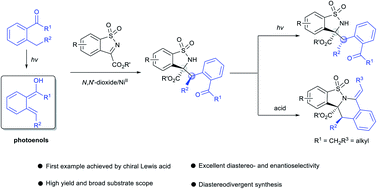
A diastereo- and enantioselective photoenolization/Mannich (PEM) reaction of ortho-alkyl aromatic ketones with benzosulfonimides was established by utilizing a chiral N,N′-dioxide/Ni(OTf)2 complex as the Lewis acid catalyst. It afforded a series of benzosulfonamides and the corresponding ring-closure products, and a reversal of diastereoselectivity was observed through epimerization of the benzosulfonamide products under continuous irradiation. On the basis of the control experiments, the role of the additive LiNTf2 in achieving high stereoselectivity was elucidated. This PEM reaction was proposed to undergo a direct nucleophilic addition mechanism rather than a hetero-Diels–Alder/ring-opening sequence. A possible transition state model with a photoenolization process was proposed to explain the origin of the high level of stereoinduction.


 Please wait while we load your content...
Please wait while we load your content...