Copper catalyzed borocarbonylation of benzylidenecyclopropanes through selective proximal C–C bond cleavage: synthesis of γ-boryl-γ,δ-unsaturated carbonyl compounds†
Abstract
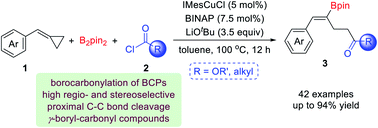
A copper catalyzed borocarbonylation of BCPs via proximal C–C bond cleavage for the synthesis of γ-boryl-γ,δ-unsaturated carbonyl compounds has been developed. Using substituted benzylidenecyclopropanes (BCPs) and chloroformates as starting material, a broad range of γ-boryl-γ,δ-unsaturated esters were prepared in moderate to excellent yields with excellent regio- and stereoselectivity. Besides, when aliphatic acid chlorides were used in this reaction, γ-boryl-γ,δ-unsaturated ketones could be produced in excellent yields. When substituted BCPs were used as substrates, the borocarbonylation occurred predominantly at the proximal C–C bond trans to the phenyl group in a regio- and stereoselective manner, which leads to the Z-isomers as the products. This efficient methodology involves the cleavage of a C–C bond and the formation of a C–C bond as well as a C–B bond, and provides a new method for the proximal C–C bond difunctionalization of BCPs.


 Please wait while we load your content...
Please wait while we load your content...