Identification of novel functional mini-receptors by combinatorial screening of split-WW domains†
Abstract
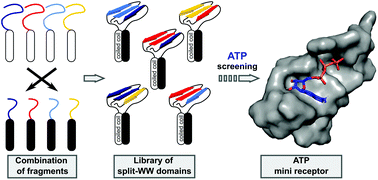
β-Sheet motifs such as the WW domain are increasingly being explored as building blocks for synthetic biological applications. Since the sequence-structure relationships of β-sheet motifs are generally complex compared to the well-studied α-helical coiled coil (CC), other approaches such as combinatorial screening should be included to vary the function of the peptide. In this study, we present a combinatorial approach to identify novel functional mini-proteins based on the WW-domain scaffold, which takes advantage of the successful reconstitution of the fragmented WW domain of hPin1 (hPin1WW) by CC association. Fragmentation of hPin1WW was performed in both loop 1 (CC-hPin1WW-L1) and loop 2 (CC-hPin1WW-L2), and the respective fragments were linked to the strands of an antiparallel heterodimeric CC. Structural analysis by CD and NMR spectroscopy revealed structural reconstitution of the WW-domain scaffold only in CC-hPin1WW-L1, but not in CC-hPin1WW-L2. Furthermore, by using 1H–15N HSQC NMR, fluorescence and CD spectroscopy, we demonstrated that binding properties of fragmented hPin1WW in CC-hPin1WW-L1 were fully restored by CC association. To demonstrate the power of this approach as a combinatorial screening platform, we synthesized a four-by-six library of N- and C-terminal hPin1WW-CC peptide fragments that was screened for a WW domain that preferentially binds to ATP over cAMP, phophocholine, or IP6. Using this screening platform, we identified one WW domain, which specifically binds ATP, and a phosphorylcholine-specific WW-based mini-receptor, both having binding dissociation constants in the lower micromolar range.


 Please wait while we load your content...
Please wait while we load your content...