Sulfur(iv)-mediated umpolung α-heterofunctionalization of 2-oxazolines†
Abstract
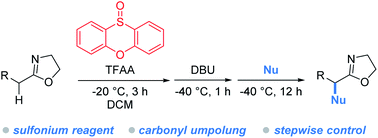
The α-umpolung of carbonyl compounds significantly expands the boundaries of traditional carbonyl chemistry. Despite various umpolung methods available today, reversing the inherent reactivity of carbonyls still remains a substantial challenge. In this article, we report the first use of sulfonium salts, in lieu of well-established hypervalent iodines, for the carbonyl umpolung event. The protocol enables the incorporation of a wide variety of heteroatom nucleophiles into the α-carbon of 2-oxazolines. The success of this investigation hinges on the following factors: (1) the use of sulfoxides, which are abundant, structurally diverse and tunable, and easily accessible, ensures the identification of a superior oxidant namely phenoxathiin sulfoxide for the umpolung reaction; (2) the “assembly/deprotonation” protocol previously developed for rearrangement reactions in our laboratory was successfully applied here for the construction of α-umpoled 2-oxazolines.
- This article is part of the themed collection: 2022 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...