Structure-mechanics statistical learning uncovers mechanical relay in proteins†
Abstract
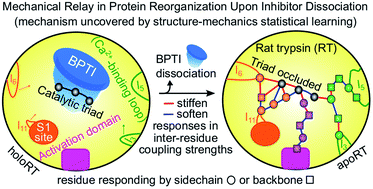
A protein's adaptive response to its substrates is one of the key questions driving molecular physics and physical chemistry. This work employs the recently developed structure-mechanics statistical learning method to establish a mechanical perspective. Specifically, by mapping all-atom molecular dynamics simulations onto the spring parameters of a backbone-side-chain elastic network model, the chemical moiety specific force constants (or mechanical rigidity) are used to assemble the rigidity graph, which is the matrix of inter-residue coupling strength. Using the S1A protease and the PDZ3 signaling domain as examples, chains of spatially contiguous residues are found to exhibit prominent changes in their mechanical rigidity upon substrate binding or dissociation. Such a mechanical-relay picture thus provides a mechanistic underpinning for conformational changes, long-range communication, and inter-domain allostery in both proteins, where the responsive mechanical hotspots are mostly residues having important biological functions or significant mutation sensitivity.
- This article is part of the themed collection: 2022 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...