Student thinking profiles within a small group addressing problems in thermodynamics as part of a physical chemistry unit: reflections for students and instructors
Abstract
This paper reports on how a group of students think when trying to solve a set of thermodynamic revision exercises in the context of collaborative small group conversation at the undergraduate level. The revision exercises involved exploring the relationship between enthalpy and temperature, entropy and temperature, and entropy of a gas under compression. Four students enrolled in a science or science teaching degree comprised the group. Two workshops, conducted about one month apart, were convened and student and instructor diaries were used to explore the thinking profiles illuminated in the diaries. The thinking profiles fitted into the categories: routine-level, object-level, process-level and meta-level. A surprisingly large amount of meta-level thinking was exhibited by the students. It was found advantageous to add adjectival qualifiers to object-level and process-level profiles to more fully interpret what the students had to say. The qualifiers were: incoherent, intuitive, algorithmic, and integrative. The workshops proved beneficial not only to the students but also to the instructor. The use of equations like 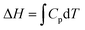 mixes the result of a definite integral symbolic format (left-hand side) with the indefinite integral symbolic format (right-hand side), and leads to students finding it difficult to distinguish between H, dH, and ΔH for enthalpy and S, dS, and ΔS for entropy. Students communicated their feelings freely and there was a general consensus within the group that the workshops were really beneficial.
mixes the result of a definite integral symbolic format (left-hand side) with the indefinite integral symbolic format (right-hand side), and leads to students finding it difficult to distinguish between H, dH, and ΔH for enthalpy and S, dS, and ΔS for entropy. Students communicated their feelings freely and there was a general consensus within the group that the workshops were really beneficial.


 Please wait while we load your content...
Please wait while we load your content...