Novel derivatives of eugenol as potent anti-inflammatory agents via PPARγ agonism: rational design, synthesis, analysis, PPARγ protein binding assay and computational studies†
Abstract
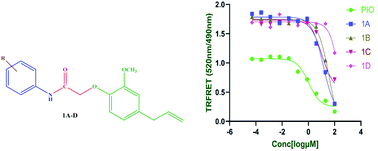
Eugenol is a natural product abundantly found in clove buds known for its pharmacological activities such as anti-inflammatory, antidiabetic, antioxidant, and anticancer activities. It is well known from the literature that peroxisome proliferator-activated receptors (PPARγ) have been reported to regulate inflammatory responses. In this backdrop, we rationally designed semi-synthetic derivatives of eugenol with the aid of computational studies, and synthesized, purified, and analyzed four eugenol derivatives as PPARγ agonists. Compounds were screened for PPARγ protein binding by time-resolved fluorescence (TR-FRET) assay. The biochemical assay results were favorable for 1C which exhibited significant binding affinity with an IC50 value of 10.65 μM as compared to the standard pioglitazone with an IC50 value of 1.052 μM. In addition to the protein binding studies, as a functional assay, the synthesized eugenol derivatives were screened for in vitro anti-inflammatory activity at concentrations ranging from 6.25 μM to 400 μM. Among the four compounds tested 1C shows reasonably good anti-inflammatory activity with an IC50 value of 133.8 μM compared to a standard diclofenac sodium IC50 value of 54.32 μM. Structure–activity relationships are derived based on computational studies. Additionally, molecular dynamics simulations were performed to examine the stability of the protein–ligand complex, the dynamic behavior, and the binding affinity of newly synthesized molecules. Altogether, we identified novel eugenol derivatives as potential anti-inflammatory agents via PPARγ agonism.


 Please wait while we load your content...
Please wait while we load your content...