Ag3VO4/g-C3N4/diatomite ternary compound reduces Cr(vi) ion in aqueous solution effectively under visible light
Abstract
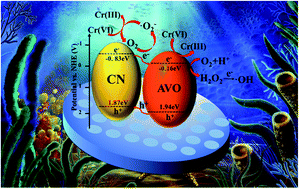
In recent years, the conversion of Cr(VI) to Cr(III) ions by semiconductor photocatalysis technology has been considered to be an effective method to solve this problem. In this paper, a kind of ternary composite, Ag3VO4/g-C3N4/diatomite (AVO/CN/DT), was synthesized by a two-step method (annealing–precipitation). Through a series of characterization analyses, the crystal morphology, microstructure, optical properties and photoelectrochemical properties of the material were characterized and analyzed. The band edge of g-C3N4 was red-shifted due to the addition of Ag3VO4 and diatomite. Consequently, the visible light response of the composites was intensified. Taking Cr(VI) in aqueous solution as a target pollutant, the degradation efficiency using 4AVO/CN/0.06DT reached 70% within 60 min under visible light irradiation, far exceeding the degradation efficiency using the pure substances. The cyclic degradation performance of the composite material was tested, and it still had a stable degradation effect after three cycles. The degradation efficiency in solution at different pH values was investigated. When the pH value of the solution gradually increased, the degradation efficiency gradually decreased, which was mainly caused by the different forms of Cr(VI) under different pH values. A corresponding degradation mechanism was proposed. Diatomite provided a reaction site for Ag3VO4 and g-C3N4, which promoted the photoreduction of Cr(VI). This work provides some reference significance for deepening the application field of diatomite and treating heavy metal ion wastewater.


 Please wait while we load your content...
Please wait while we load your content...