Direct activation of alcohols via perrhenate ester formation for an intramolecular dehydrative Friedel–Crafts reaction†
Abstract
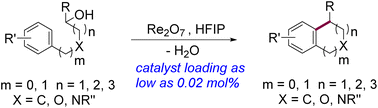
This manuscript describes a general method for the intramolecular dehydrative Friedel–Crafts reaction of alcohols catalyzed by Re2O7 in hexafluoroisopropanol (HFIP). Under the optimized conditions, δ-aryl alcohols react with high efficiency to give various pharmaceutically significant organic scaffolds, such as tetrahydronaphthalene, tetrahydroquinoline, tetrahydroisoquinoline, chromane, and isochromane derivatives. The potential application of this methodology was exemplified by the formal or total synthesis of several commercially available drugs or biologically relevant molecules, including sertraline, nafenopin, etc. DFT calculations showed the preference for the activation of hydroxyl groups by Re2O7via perrhenate ester formation followed by heterolytic C–O bond cleavage over Brønsted acid-catalyzed dehydration.


 Please wait while we load your content...
Please wait while we load your content...