Direct C(sp3)–H difluoromethylation via radical–radical cross-coupling by visible-light photoredox catalysis†
Abstract
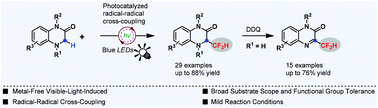
Herein, a radical–radical cross-coupling strategy for direct difluoromethylation of the C(sp3)–H bond is reported. This transformation was readily accomplished under transition metal-free photoredox catalysis in the presence of 3 mol% of an organic photocatalyst, allowing the direct difluoromethylation of C(sp3)–H of a wide variety of 1,4-dihydroquinoxalin-2-ones in good yields under mild reaction conditions. Moreover, various 3-difluoromethyl quinoxalin-2-ones were also easily furnished in a one-pot manner through this radical difluoromethylation protocol. Mechanistic studies clearly reveal that the radical–radical cross-coupling between a difluoromethyl radical and a C(sp3) radical is responsible for this transformation. To the best of our knowledge, this is the first example of radical–radical cross-coupling difluoromethylation of the C(sp3)–H bond, which not only provides a promising strategy for a straightforward installation of a CF2H group at a C(sp3) center but would also promote the development of other new fluorination and fluoroalkylation methods.
- This article is part of the themed collection: FOCUS: Radical-involved chemical transformations


 Please wait while we load your content...
Please wait while we load your content...