Isomeric effect of naphthyl spacers on structures and properties of isostructural porous crystalline frameworks†
Abstract
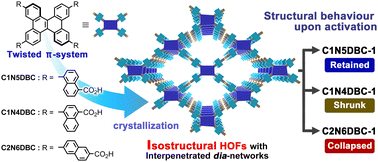
In reticular chemistry, many studies have attempted to design a series of isostructural frameworks with specific structures and properties by manipulating the constituent molecules. Frameworks constructed through non-covalent interactions, such as hydrogen-bonded organic frameworks (HOFs), are highly sensitive to subtle molecular structural differences, but a systematic study of them has not been done. The present study describes the construction of three types of isostructural, dia-topological HOFs (C1N4DBC-1, C1N5DBC-1, and C2N6DBC-1) from dibenzo[g,p]chrysene (DBC) derivatives (C1N4DBC, C1N5DBC, and C2N6DBC) that possess 1,4-, 1,5-, and 2,6-substituted carboxynaphthyl groups, respectively. The frameworks were formed using directional H-bonds between carboxy groups and the shape-fitted docking of twisted DBC cores. However, despite the isostructural frameworks, they exhibited different dynamic behaviours toward guest removal, with C1N4DBC showing pore shrinkage, C1N5DBC retaining its structure, and C2N6DBC collapsing to an amorphous state. Furthermore, in situ PXRD measurements with benzene vapour adsorption/desorption showed that C1N4DBC underwent pore-breathing behaviour through crystal-to-crystal transition. These differences are attributed to the pore geometry and hydrogen bonding style due to the isomeric effect, which is an essential principle for constructing the desired HOFs.
- This article is part of the themed collection: 2023 Materials Chemistry Frontiers HOT articles


 Please wait while we load your content...
Please wait while we load your content...