Sodium site occupancy and phosphate speciation in natrophosphate are invariant to changes in NaF and Na3PO4 concentration†
Abstract
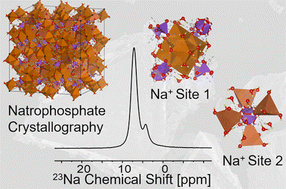
Recent interest in the crystal structure of natrophosphate (Na7FPO4·19H2O) has sought to better understand the propensity of this phase to vary in composition through either alteration of the fluoride/phosphate ratio or subspeciation of phosphate (PO43−) into hydrogen phosphate (HxPO43−x). To address questions brought up in the analysis of the solubility of natrophosphate in multicomponent NaF : Na3PO4 : H2O systems relevant to fundamental studies of select geological deposits and industrial processes pertinent to radioactive waste, this work deploys X-ray diffraction, scanning electron microscopy, Raman spectroscopy, Fourier transform infra-red spectroscopy, solid-state multinuclear (1H, 31P, 19F, and 23Na) Magic Angle Spinning Nuclear Magnetic Resonance (MAS NMR) spectroscopy and 23Na multiple quantum MAS NMR spectroscopy. Scrutiny of the integrated microscopy, diffraction and spectroscopy results indicate that the crystalline structure and stoichiometry of natrophosphate are invariant to changes in the fluoride/phosphate ratio of the solution and that formation of hydrogen phosphate can be neglected. This improved knowledge of the chemical composition of natrophosphate crystallites will aide in the application of solubility models relevant to geological studies and radioactive waste processing.


 Please wait while we load your content...
Please wait while we load your content...