Expedient synthesis and ring-opening metathesis polymerization of pyridinonorbornenes†
Abstract
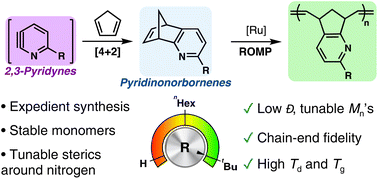
Pyridine-containing polymers are promising materials for a variety of applications from the capture of contaminants to the self-assembly of block copolymers. However, the innate Lewis basicity of the pyridine motif often hampers living polymerization catalyzed by transition-metal complexes. Herein, we report the expedient synthesis of pyridinonorbornene monomers via a [4 + 2] cycloaddition between 2,3-pyridynes and cyclopentadiene. Well-controlled ring-opening metathesis polymerization was enabled by careful structural design of the monomer. Polypyridinonorbornenes exhibited high Tg and Td, a promising feature for high-temperature applications. Investigation of the polymerization kinetics and of the reactivity of the chain ends shed light on the influence of nitrogen coordination on the chain-growth mechanism.
- This article is part of the themed collection: Polymer Chemistry Emerging Investigators Series


 Please wait while we load your content...
Please wait while we load your content...