Enhancing the performances of physically cross-linked photodeformable main-chain azobenzene poly(ester-amide)s via chemical structure engineering†
Abstract
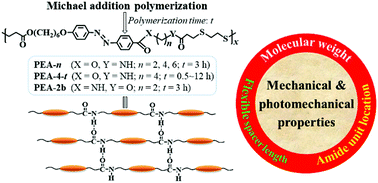
The synthesis of a series of photodeformable main-chain azobenzene (azo) poly(ester-amide)s (PEAs) with different flexible spacer lengths, molecular weights, and amide unit location via Michael addition polymerization and study of the correlation between their chemical structures and performances are first described. These PEAs showed low crystallinity degrees and glass transition temperatures, high thermal stability, reversible photoresponsivity, and amide unit-induced hydrogen bonding interactions. Their chemical structures exhibited a large influence on their film-forming capability as well as mechanical and photomechanical properties. Physically cross-linked uniaxially oriented fibers and films with high mechanical strength and reversible room-temperature photoinduced bending/unbending behaviors were fabricated from high-molecular-weight PEAs with a flexible spacer (–(CH2)n–) between the amide unit and azo mesogen (PEA-n), whereas their low-molecular-weight counterpart and PEA with the amide unit directly connected to the azo mesogen (PEA-2b) only formed brittle fibers/films. The mechanical strength of PEA-n fibers/films increased with the increase in the molecular weights and flexible spacer length, whereas optimal photomechanical effects were observed for PEA-n samples with a moderate molecular weight and a shorter flexible spacer (n = 2). The photomobile rates and amplitudes of PEA fibers/films can be well correlated with their elastic moduli and photoinduced stress. In addition, the decisive role of the aggregation states of PEA samples in their mechanical strength as well as those of both the amide unit-induced physical cross-linking and alignment structure in their photomobility was also demonstrated. This work not only presents photodeformable main-chain azo PEAs with enhanced performances via chemical structure engineering, but also lays the foundation for rationally designing more advanced physically cross-linked photomobile main-chain azo polymers.


 Please wait while we load your content...
Please wait while we load your content...