Transition-metal-free radical difluorobenzylation/cyclization of unactivated alkenes: access to ArCF2-substituted ring-fused quinazolinones†
Abstract
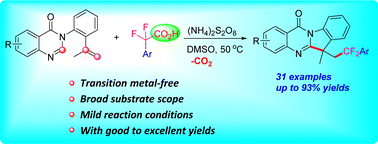
A mild and efficient transition-metal-free radical difluorobenzylation/cyclization of unactivated alkenes toward the synthesis of difluorobenzylated polycyclic quinazolinone derivatives with easily accessible α,α-difluoroarylacetic acids has been developed. This transformation has the advantages of wide functional group compatibility, a broad substrate scope, and operational simplicity. This methodology provided a highly attractive access to pharmaceutically valuable ArCF2-containing polycyclic quinazolinones.


 Please wait while we load your content...
Please wait while we load your content...