Abstract
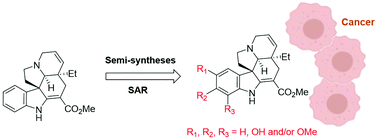
We demonstrated here a series of Aspidosperma terpenoid alkaloids can be quickly prepared using semisynthesis from naturally sourced tabersonine, featuring multiple oxygen-based substituents on the indole ring such as hydroxy and methoxy groups. This panel of complex compounds enabled the exploration of indole modifications to optimize the indole alkaloids’ anticancer activity, generating lead compounds (e.g., with C15-hydroxy, C16-methoxy, and/or C17-methoxy derivatizations) that potently inhibit cancer cell line growth in the single-digit micromolar range. These results can help guide the development of Aspidosperma terpenoid alkaloid therapeutics. Furthermore, this synthetic approach features late-stage facile derivatization on complex natural product molecules, providing a versatile path to indole derivatization of this family of alkaloids with diverse chemical functionalities for future medicinal chemistry and chemical biology discoveries.


 Please wait while we load your content...
Please wait while we load your content...