Chemoenzymatic total synthesis of nodulones C and D using a naphthol reductase of Magnaporthe grisea†
Abstract
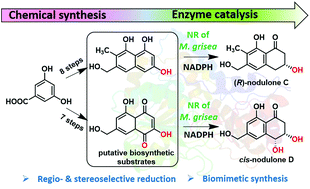
Herein, the asymmetric and chemoenzymatic synthesis of (R)-nodulone C, cis-nodulone D and related (R)-dihydronaphthalenone is reported. It involves multistep chemical synthesis of putative biosynthetic substrates followed by regio- and stereoselective reduction using a NADPH-dependent tetrahydroxynaphthalene reductase of Magnaporthe grisea to obtain chiral nodulones in a biomimetic fashion.


 Please wait while we load your content...
Please wait while we load your content...