An overview of metal-free sustainable nitrogen-based catalytic knoevenagel condensation reaction
Abstract
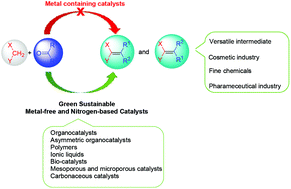
Knoevenagel condensation reaction counts as a vital condensation in organic chemistry due to the synthesis of valuable intermediates, heterocycles, and fine chemicals from commercially available reactants through forming new C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds between an aldehyde or ketone and active methylene compounds. Therefore, the catalytic Knoevenagel condensation reaction has continuously received significant interest in different aspects, i.e., investigating the catalytic efficiency of new heterogeneous and homogeneous catalysts. Furthermore, metal-free catalysis has recently attracted considerable attention because of environmental concerns. This review summarizes the most recent studies on utilizing metal-free nitrogen-based catalysts in the Knoevenagel condensation reaction, including organocatalysts, polymers, ionic liquids, and bio- and carbon-based catalysts. The substrate scope, the optimal reaction conditions, selectivity, the desired product yield, merits, and limitations of each method are discussed. In addition, the recyclability, biocompatibility, and biodegradability of catalysts are reported. This review also covers the approaches that influence the selectivity of the Knoevenagel products.
C bonds between an aldehyde or ketone and active methylene compounds. Therefore, the catalytic Knoevenagel condensation reaction has continuously received significant interest in different aspects, i.e., investigating the catalytic efficiency of new heterogeneous and homogeneous catalysts. Furthermore, metal-free catalysis has recently attracted considerable attention because of environmental concerns. This review summarizes the most recent studies on utilizing metal-free nitrogen-based catalysts in the Knoevenagel condensation reaction, including organocatalysts, polymers, ionic liquids, and bio- and carbon-based catalysts. The substrate scope, the optimal reaction conditions, selectivity, the desired product yield, merits, and limitations of each method are discussed. In addition, the recyclability, biocompatibility, and biodegradability of catalysts are reported. This review also covers the approaches that influence the selectivity of the Knoevenagel products.


 Please wait while we load your content...
Please wait while we load your content...