Fine-tuning the acceptor–donor ability of star shaped triarylborane–triphenylamine conjugates: synthesis, characterization and anion binding studies†
Abstract
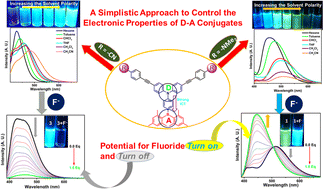
The design and facile synthesis of three new star shaped triarylborane-triphenyl amine donor–acceptor systems 1, 2 and 3 are reported with their characterization. The optical properties were studied taking into account their solid-state emitting and solvatochromic behaviours. The compounds showed similar absorption as well as emission feature in dichloromethane solutions. However, compound 1 showed two emission bands which correspond to the charge transfer bands (ICT) and local excited state emission, whereas compounds 2 and 3 showed only one emission band, i.e., the ICT band. The ICT process is seen to be more predominant in 1 as compared with 2 and 3 due to the strong electron donating tendency of –NMe2 in 1. The solvatochromic behaviour of compounds 1–3 was studied, which shows a bathochromic shift in the emission spectra with increasing solvent polarity which is an ideal behaviour of donor–acceptor systems exhibiting the ICT process. Furthermore, the compounds showed high bright emission in the solid state in the region of 550, 495, and 475 nm respectively. The anion binding studies were carried out, which revealed that compounds 1–3 showed high selectivity and sensitivity towards the fluoride ion even in the presence of other competitive anions, with compound 1 showing fluorescence enhancement and 2 and 3 showing fluorescence quenching in the presence of fluoride. Moreover, the binding constants, quenching efficiency and detection limit values are found to be slightly higher or similar to that of known triarylborane based fluoride receptors.


 Please wait while we load your content...
Please wait while we load your content...