A Ni-MOF based luminescent sensor for selective and rapid sensing of Fe(ii) and Fe(iii) ions†
Abstract
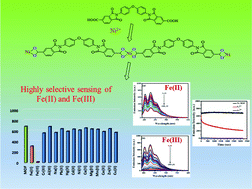
A V-shaped, bis(trimellitimide) linker in the metal organic framework (MOF) family was prepared and further used to synthesize a novel nickel-based luminescent MOF. The linker as well as the MOF were further characterized by various instrumental techniques. Furthermore, the MOF's luminescent properties were studied for selective detection of Fe2+ and Fe3+ through fluorescence quenching. As one may know, Fe2+ is essential for oxygen transport and photosynthesis, which are controlled by oxidases and reductases. Fe3+, on either arm, is harmful because of its persistent binding to oxygen. As a result, in the presence of Fe2+, precise Fe3+ detection from aqueous systems is crucial. Unfortunately, due to their molecular resemblance, it is difficult to distinguish them accurately. The synthesized Ni-MOF is highly sensitive to Fe3+ and has a very quick fluorescence reaction time, allowing for rapid Fe2+ and Fe3+ distinction. Here, the limit of detection (LOD) was found to be 25.03 μM and 15.44 μM with quenching efficiencies of 54% and 98% for Fe2+ and Fe3+, respectively. The introduction of Fe2+ and Fe3+ effectively quenches the fluorescence emission observed for the Ni-MOF more effectively by offering a rapid response, good selectivity, and sensitivity over other metal ions.


 Please wait while we load your content...
Please wait while we load your content...