Quantitative structure–activity relationship modeling of the inhibitory activities of sulfonylurea herbicides and proposition of new derivatives
Abstract
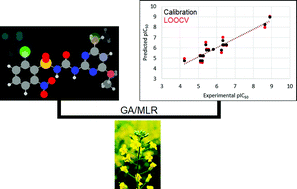
The quantitative structure–activity relationship study of the herbicidal activity of a series of sulfonylurea derivatives against Brassica napus is reported herein. A few multivariate image analysis (MIA) descriptors have been selected through a genetic algorithm from a pool of pixels renumbered to encode atomic properties, namely the van der Waals radii and electronegativity. These descriptors correlate with the B. napus growth inhibition to a great extent (r2 = 0.90–0.91, q2 = 0.76–0.82) and, therefore, could be used to estimate the pIC50 values of hitherto unknown sulfonylurea analogs (IC50 = half maximal inhibitory concentration). The design of these novel derivatives was guided by MIA contour maps that reveal the substituent effects on the response variable. As a result, four agrochemical candidates were proposed and three of them exhibited calculated pIC50 values higher than that of chlorsulfuron and the data set compounds. This may be an important step to drive the synthesis of new sulfonylurea herbicides with improved properties against resistant weeds.


 Please wait while we load your content...
Please wait while we load your content...