The doping of B in ZnO/CdS for enhanced visible-light H2 production†
Abstract
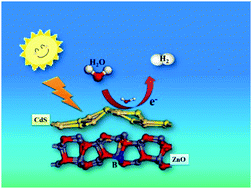
The visible-light water splitting enhancement of B in ZnO/CdS was systematically studied and the B-doped ZnO/CdS rods (B-ZnO/CdS) showed an excellent H2 generation rate of 13 100 μmol h−1 g−1, mostly 2 times higher than that of ZnO/CdS (ca. 5249 μmol h−1 g−1, λ > 420 nm), indicating that due to the introduction of the B element, the electron separation efficiency in ZnO/CdS was improved significantly. Through various characterization methods (e.g., PL spectroscopy, UV-Diffuse reflection spectroscopy and photoelectric response), the enhanced electron separation efficiency in the B-ZnO/CdS system was directly demonstrated. In addition, according to the density functional theory (DFT) calculation results, B-ZnO/CdS exhibited a negative adsorption energy of water (EH2O* = −1.22 eV), and the d-band center of the Cd atom had a positive shift from −8.46 to −8.45 eV, which was favorable for water absorption and dissociation. From the charge density difference and density of states, it could be found that electrons tend to transfer from ZnO to H2O at the Cd active sites. The accumulated electrons in H2O could extend the H–OH bond length from 0.975 Å to 0.983 Å, thereby facilitating the dissociation of H2O.


 Please wait while we load your content...
Please wait while we load your content...