A pyridine based Schiff base as a selective and sensitive fluorescent probe for cadmium ions with “turn-on” fluorescence responses†
Abstract
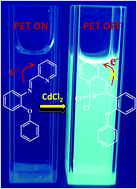
A new fluorescent probe based on a pyridine Schiff base (2-(pyridin-2-ylmethylene)phenoxyaniline, PMPA) working with the PET mechanism and chelation enhanced fluorescence (CHEF) was designed and synthesized for the selective detection of cadmium ions. Probe PMPA exhibited a turn-on fluorescence response toward Cd2+ with a wide pH range (pH = 5–9), a rapid response time (3 minutes), excellent sensitivity (LOD = 0.12 μM) and good reversibility. The binding constant (K) of PMPA with Cd2+ was calculated to be 4.36 × 104 M−1. The 1 : 1 stoichiometry of the complex between PMPA and Cd2+ ions was confirmed through Job's plot and ESI-MS and 1H/13C NMR spectra, and further confirmed by the X-ray crystal structure of the complex PMPA–CdCl2. Density functional theory (DFT) calculations verified the mechanism of fluorescence enhancement. Moreover, PMPA was also successfully used to detect Cd2+ in the samples (tap, pond and Hun River water samples) with satisfactory results.


 Please wait while we load your content...
Please wait while we load your content...