Novel approach for the synthesis of chiral organometallic complexes – first series of lithium 2-amino-indenide ligands bearing pendant donor groups and a unique helical bent-zirconocene†
Abstract
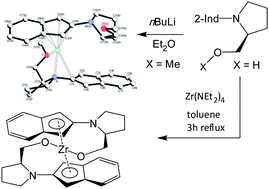
A broad series of mono- and bis-2-aminoindenyl ligands, bearing pendant donor sec-amine groups was synthesized via the reaction of indan-2-one with the corresponding secondary mono- and bis-amines: 1-H from pyrrolidine, 2-H from piperidine, 3-H from morpholine, 4-H from thiomorpholine, 5-H from dibenzylamine, 6-H2 from N,N′-diethyldiaminopropane-1,3, 7-H2 from piperazine, 8-H2 from S-(+)-prolinol, 9-H from O-methyl-S-(+)-prolinol, 10-H from O-ethyl-S-(+)-prolinol and 11-H2 from R-(-)-N-(2-anilinomethyl)-pyrrolidine. Direct metallation of 3-H, 4-H and 9-H by nBuLi led to the formation of the corresponding mono-lithiated species 3-Li, 4-Li and 9-Li. Trans-Metallation of 4-Li by ZrCl4 led to the formation of the presumably monomeric [(η5-4)ZrCl3] 12. Partial hydrolysis of 12 occurred after several attempts to crystallize it and led to the formation of its 1 : 1 adduct with the free ligand 4-H, [(η5-4)ZrCl3(μ-N,S-4-H)] 13. Direct metallation of 8-H2 with [Zr(NEt2)4] led to the formation of a chiral bent-zirconocene with a helical structure [(η5,μ-O-8)2Zr] 14. The molecular structure of enantiomerically pure 9-Li was determined using X-ray crystallographic studies. Alternating Li+ and chelating indenyl anions are linked in infinite chains. The coordination environment of Li+ consists of the η5-bonded five-membered ring of the indenyl fragment, the η1-bonded adjacent ligand and the donor sp3 nitrogen and oxygen atoms of the chelating side chain.


 Please wait while we load your content...
Please wait while we load your content...