Effect of oxygen vacancies on the electronic structure and electrochemical performance of MnMoO4: computational simulation and experimental verification
Abstract
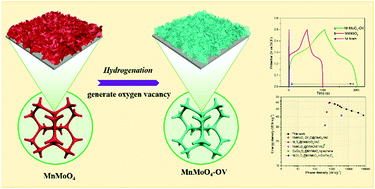
Porous nanopetals of MnMoO4 with oxygen vacancies were prepared by hydrothermal synthesis and a hydrogenation reduction method. MnMoO4–OV porous nanopetals have a higher specific surface area together with a more diverse pore size distribution than MnMoO4 nanopetals. The density functional theory calculation results reveal that MnMoO4–OV has a lower energy band gap (0.642 eV) than MnMoO4 (1.095 eV). The electronic density of states plots reveal that MnMoO4–OV displays more electron state distribution at the Fermi energy level than MnMoO4. The MnMoO4–OV porous nanopetals achieve a higher specific capacitance (1920 g−1) than MnMoO4 (921 F g−1) at 1 A g−1. They also achieve a superior capacitance retention rate (85.5%) to that of MnMoO4 (69.1%) with the current density increasing from 1 to 10 A g−1. The introduction of oxygen vacancies can increase the carrier density, accelerate the electron transfer, enhance the electrical conductivity, and accordingly strengthen the redox reactivity of MnMoO4–OV. An asymmetric supercapacitor was also constructed by using MnMoO4–OV as the positive electrode and activated carbon as the negative electrode. It achieves a high energy density of 59.8 W h kg−1 at a power density of 750 W kg−1, together with a good cycle life. Both experimental measurements and theoretical calculations were conducted to prove the promotive role of oxygen vacancies in achieving the supercapacitive performance of MnMoO4–OV.


 Please wait while we load your content...
Please wait while we load your content...