The role of hydrogen bonding in tuning CEST contrast efficiency: a comparative study of intra- and inter-molecular hydrogen bonding†
Abstract
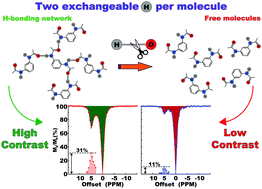
The effectiveness of MRI as a diagnostic tool has increased tremendously after the discovery of contrast agents (CAs). Most of the clinically approved CAs at present are relaxation-based and their containing metal raises safety concerns at times for in vivo applications. The search for a safer alternative led to the discovery of Chemical Exchange Saturation Transfer (CEST)-based CAs. Many of the CEST CAs, particularly the diamagnetic (diaCEST) ones, use exchangeable hydroxy and amide functional groups that are known to form hydrogen-bond networks in solution. As hydrogen bonding alters the lability of the participating proton groups, it is expected that hydrogen bonding will directly influence the efficiency of diaCEST CAs. However, very few reports are found on systematic and comparative studies of the role of hydrogen bonding in this context. In this work, we compared the CEST properties of isomers of diacetamide with varying hydrogen bonding capabilities. We found that hydrogen bonding not only influences the efficiency but its presence is pivotal for the mere existence of these CAs. Through a concerted temperature-dependent measurement of the translational diffusion coefficient, exchange coefficient and CEST efficiency, we showed that at mild heating above the physiological temperature, when the hydrogen-bonding network starts breaking apart the CEST efficiency drops rapidly in spite of an increase in the exchange constant. We also found that, with all other parameters being the same, inter- and intra-molecular hydrogen-bonding influence the CEST efficiency quite differently.


 Please wait while we load your content...
Please wait while we load your content...