Understanding tyrosine self-assembly: from dimer assembly to magnetized fluorescent nanotubes embedded into PVA films†
Abstract
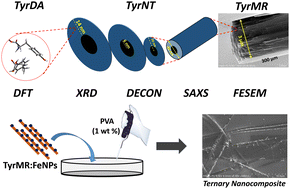
Biomolecular self-assembly has emerged as a remarkable tool for the development of functional materials at biological interfaces. Considerable attention has been focused on amino acid-based tubular structures, such as protein (tubulin) microtubes and β-sheets containing diphenylalanine nanotubes, for their role in cognitive function and neurodegenerative disorders. Further, the unique photophysical properties displayed by single amino acid self-assembled structures may find novel applications in the field of bio-optoelectronics and biophotonics. However, the nanoscale crystallinity and its role in hierarchical self-assembly formation may lead to a distinctive path for a particular molecular system. In the quest to understand tyrosine nanoscale self-assembly, we have demonstrated that tyrosine forms microrod (TyrMR) structures via the multiple stacking of tyrosine nanotubes (TyrNTs), which are composed of tyrosine dimer assemblies (TyrDAs) as the basic unit. Further, we showed that the unique photoluminescence displayed by TyrMR structures can be tuned via the co-assembly of phenylalanine and thioflavin T dye. The photoactive TyrMRs were magnetized using ferrite nanoparticles to generate a binary nanocomposite, which was further embedded into a PVA thin film to produce a magneto-opto-active ternary nanocomposite. The fabricated ternary nanocomposite structure presented multifunctional features, such as nanocrystallinity, tunable photoluminescence, and a magnetic response, which may pave the way for the development of integrated functional materials with potential applications in multimodal optical imaging, bioimaging, and biophotonics.


 Please wait while we load your content...
Please wait while we load your content...