Interstitial flow enhances the formation, connectivity, and function of 3D brain microvascular networks generated within a microfluidic device†
Abstract
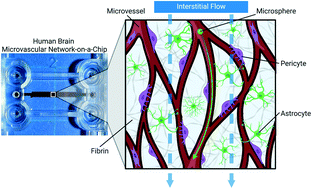
The bulk flow of interstitial fluid through tissue is an important factor in human biology, including the development of brain microvascular networks (MVNs) with the blood–brain barrier (BBB). Bioengineering perfused, functional brain MVNs has great potential for modeling neurovascular diseases and drug delivery. However, most in vitro models of brain MVNs do not implement interstitial flow during the generation of microvessels. Using a microfluidic device (MFD), we cultured primary human brain endothelial cells (BECs), pericytes, and astrocytes within a 3D fibrin matrix with (flow) and without (static) interstitial flow. We found that the bulk flow of interstitial fluid was beneficial for both BEC angiogenesis and vasculogenesis. Brain MVNs cultured under flow conditions achieved anastomosis and were perfusable, whereas those under static conditions lacked connectivity and the ability to be perfused. Compared to static culture, microvessels developed in flow culture exhibited an enhanced vessel area, branch length and diameter, connectivity, and longevity. Although there was no change in pericyte coverage of microvessels, a slight increase in astrocyte coverage was observed under flow conditions. In addition, the immunofluorescence intensity of basal lamina proteins, collagen IV and laminin, was nearly doubled in flow culture. Lastly, the barrier function of brain microvessels was enhanced under flow conditions, as demonstrated by decreased dextran permeability. Taken together, these results highlighted the importance of interstitial flow in the in vitro generation of perfused brain MVNs with characteristics similar to those of the human BBB.


 Please wait while we load your content...
Please wait while we load your content...