Determining 88Sr/86Sr of barite using the Na2CO3 exchange method†
Abstract
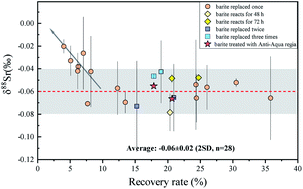
This study presents a method of high-precision stable Sr isotope measurement for barite using the Na2CO3 exchange reaction and MC-ICP-MS. The reliability of this method was strictly tested by measurement of the synthetic barite standard. The synthetic barites were made by mixing the natural barite standard GBW07814 with three strontium-rich matrix rocks (carbonate JLs-1, andesite AGV-2, and black shale SGR-1) in different proportions. The average 87Sr/86Sr and δ88Sr of synthetic barites are 0.713556 ± 0.000013 (2SD, n = 22) and −0.07 ± 0.02‰ (2SD, n = 22), respectively, which are consistent with the values of GBW07814 (87Sr/86Sr of 0.713555 ± 0.000023, 2SD, n = 46 and δ88Sr of −0.06 ± 0.02‰, 2SD, n = 28). High recovery in the exchange reaction was achieved by using excessive Na2CO3 to avoid Sr isotope fractionation. Furthermore, the potential matrix effects in chemical purification were rigorously evaluated by analyzing the synthetic solution made by mixing SRM987 and matrix elements. The average δ88Sr of the synthetic solution is −0.01 ± 0.04‰ (2SD, n = 4), consistent with the δ88Sr of SRM987 (0.00‰). Based on long-term analyses of USTC-Sr (pure Sr solution), the external precision of δ88Sr was found to be better than 0.03‰ (2SD, n = 92). We further measured the 87Sr/86Sr and δ88Sr of five natural barite standards (GBW07811, GBW07812, GBW07814, GBW07815 and GBW07816), revealing significant stable Sr isotope fractionation in natural barites by 0.37‰.


 Please wait while we load your content...
Please wait while we load your content...