Utilization of ionic liquids for preferential separation of thorium during the determination of trace metallic constituents in thorium matrix using ICP-OES
Abstract
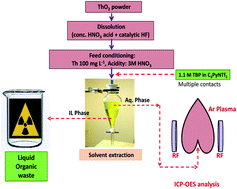
To avoid spectral interference it is required to separate Th preferentially during the determination of trace metallic constituents from Th matrix using ICP-OES. It was demonstrated that ionic liquids can be successfully applied for the removal of emission-rich thorium matrix below tolerance limit without any compromise in estimation and other analytical capabilities during trace metal assay in thorium-based fuel samples using ICP-OES. Ionic liquid-based ‘green’ separation methodology was used for the drastic reduction in organic phase requirement, generation of organic liquid waste, time of analysis, personal exposure towards the radiotoxic environment, fewer chances of contamination due to simplified sample processing. Thorium was found to form neutral Th(NO3)4·3 TBP predominantly, following the ‘solvation’ mechanism. The methodology was optimized by understanding the behaviour of analytes during chemical separation, appropriate feed conditioning and deciding the optimum ligand concentration, identification of analytical lines free from thorium interference and their analytical performance in optimized experimental and instrumental conditions. A comparative investigation was also performed using conventional separation methodology using a 1.1 M TBP/n-dodecane/3 M HNO3 system. The % recovery of the analytes was assessed. Statistical tests (Student's t-test and F test) were performed to understand the reliability of the estimation data and experimentally observed variance compared to the conventional technique.


 Please wait while we load your content...
Please wait while we load your content...