Dehydration of primary amides to nitriles in water. Late-stage functionalization and 1-pot multistep chemoenzymatic processes under micellar catalysis conditions†
Abstract
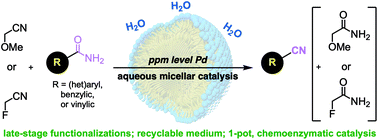
Dehydration of primary amides to the corresponding nitriles can be performed in aqueous micelles, without need of significant volumes of organic co-solvent. A variety of substrates, including late-stage functionalized molecules, can be converted utilizing an inexpensive, “sacrificial” nitrile which can accept an equivalent of water. Catalyst loadings of Pd(OAc)2, as low as 0.2–0.8 mol%, are possible under these conditions in a completely recyclable medium exhibiting very low E Factors. This process is also amenable to 1-pot, chemoenzymatic catalysis exemplifying the benefits of chemistry in water.


 Please wait while we load your content...
Please wait while we load your content...