Pathways of ammonia electrooxidation on nickel hydroxide anodes and an alternative route towards recycled fertilizers†
Abstract
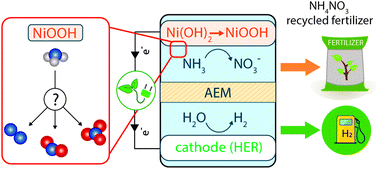
The anaerobic digestion (AD) of N-enriched waste streams combined with nutrient recovery from AD effluents represents a cost-effective waste treatment technology for simultaneous production of biogas and recycled fertilizers. Herein, we demonstrate an efficient route towards S- or P-containing NH4NO3-based fertilizers via Ni(OH)2-catalyzed electrochemical oxidation of ammonia (AOR) to NH4NO3 in 0.1 M K2SO4 and 0.1 M K2HPO4 electrolytes. In this study, we examine how operating parameters, including the electrolyte, pH, applied potential, ammonia concentration, and temperature affect the AOR product distribution at Ni(OH)2 anodes. We show that under optimized conditions concentrated ammonia solutions can be efficiently converted into NH4NO3 with up to 72% faradaic efficiencies and up to 98% ammonia removal. Based on our extensive experimental data, we propose detailed mechanisms for the formation of major products (dinitrogen, nitrite, and nitrate) and for the first time experimentally demonstrate that ammonia-to-nitrite oxidation proceeds spontaneously on NiOOH, while the nitrite-to-nitrate step is purely electrochemical. The results of this work are expected to enable integration of AOR into AD of N-containing waste, thereby enabling a promising nutrient recovery strategy for building a circular economy.


 Please wait while we load your content...
Please wait while we load your content...