Experimental study and quantum chemical calculation of free radical reactions in ciprofloxacin degradation during the UV/chlorine oxidation process†
Abstract
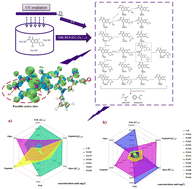
The UV/chlorine process was utilized to degrade a common quinolone, ciprofloxacin, in water. Ciprofloxacin removal was high at 92.2% in 100 s, with a first-order rate constant of 0.0026 s−1 during the process. The degradation of ciprofloxacin was found to be positively related to UV intensity and chlorine dosage and negatively related to ammonia concentration. According to FMO, MSPA, CDFT, and topological quantum chemical analysis, the N6 or N7 atoms are the most vulnerable to electrophilic or radical attacks in the piperazine ring ciprofloxacin structure, indicating the most likely reactive sites for the initial reaction. 21 degradation products were identified, including 19 products by HRMS LC–MS and 2 by GC–MS. These intermediates were mainly produced through the following three pathways: the piperazine ring's cleavage, hydroxyl addition, and defluorination reaction. Both the acute toxicity tests and ECOSAR prediction illustrated that the ecological risks cannot be effectively removed due to the production of more toxic intermediates during the degradation process.


 Please wait while we load your content...
Please wait while we load your content...