Nano-biochar modulates the formation of iron plaque through facilitating iron-involved redox reactions on aquatic plant root surfaces†
Abstract
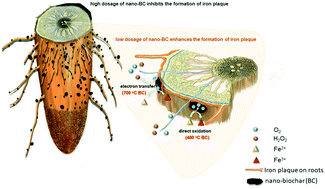
The ubiquitous presence of submicron-sized black carbon (BC) in the water environment makes it inevitably interact with root surfaces of aquatic plants. However, the nexus between catalytic ability of attached BC and iron plaque (IP) formation is not fully understood. Here, the impact of nanoscale BC particles (nano-BC) produced at 400 and 700 °C on Fe(II) oxidation and IP formation (i.e., Fe(III)-(oxyhydr)oxide coating) on root surfaces of rice seedlings was investigated via hydroponic culture. Results suggest that the formation of IP was considerably regulated by the concentration of nano-BC attached on roots, where low concentration of nano-BC (10 mg L−1 for nano400 and 5 mg L−1 for nano700) significantly (p < 0.05) enhanced the formation of IP due to Fe(II) direct oxidation/electron transfer while excess nano-BC not only impeded the formation of IP but also significantly reduced (p < 0.05) plant growth and nutrient uptake. The critical concentration of nano-BC was highly dependent on its redox potential stemming from both redox-active groups and graphitic structures in nano-BC. The newly formed BC–IP composite exhibited distinct configuration relative to the common IP, where both poorly crystalline Fe(III) (oxyhydr)oxides and Fe-containing nanoparticles were observed. Our findings provide new insights into the nano-BC facilitated formation of IP and its critical role in the growth of aquatic plants.
- This article is part of the themed collection: Environmental fate of nanomaterials


 Please wait while we load your content...
Please wait while we load your content...