Decoupling of ion pairing and ion conduction in ultrahigh-concentration electrolytes enables wide-temperature solid-state batteries†
Abstract
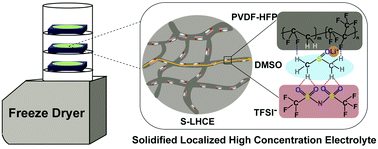
Fast ion conduction and stable interfaces are predicted to be important in the development of new electrolytes. However, many unconventional solvents for electrolytes remain challenging, although they are fast ion carriers, because of the narrow voltage window of solvent oxidation and reduction. Here, we report a general solidified localized high-concentration electrolyte (S-LHCE) strategy with the decoupling of ion pairing and ion conduction to achieve the application of unstable solvents (dimethyl sulfoxide, DMSO) in high-voltage lithium-metal batteries. By decoupling electrolytes with a non-solvating solid framework, the interfacial compatibility was further improved with lithium anodes and high-voltage cathodes. The anion migration was limited with a high Li+ transference number of 0.72, and the lithium-ion conduction was enhanced (0.27 mS cm−1 at 20 °C) by the regulated solvation structure in an ultrahigh salt concentration regime. The S-LHCE strategy enabled solid-state lithium-metal batteries with excellent electrochemical performance over a wide temperature range from −10 to 100 °C and with 83.3% and 60.1% capacity retention of the theoretical capacity when cycled at a 30C rate and a 50C rate at an evaluated temperature. The results in this work provide new insight for the application of potential but unconventional active components to high-performance electrolytes.
- This article is part of the themed collection: The EES Family Board Members Collection


 Please wait while we load your content...
Please wait while we load your content...