Synthesis and reactivity of an iridium complex based on a tridentate aminophosphano ligand†
Abstract
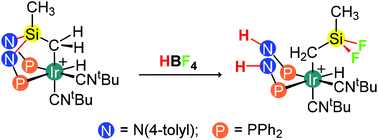
The iridium(III) hydride compound [IrH{κ3C,P,P′-(SiNP–H)}(CNtBu)2][PF6] (1PF6) was obtained by reaction of [Ir(SiNP)(cod)][PF6] with CNtBu as the result of the intramolecular oxidative addition of the SiCH2–H bond to iridium(I) [SiNP = Si(CH3)2{N(4-tolyl)PPh2}2, SiNP–H = CH2Si(CH3){N(4-tolyl)PPh2}2]. The mechanism of the reaction was investigated by NMR spectroscopy and DFT calculations showing that the pentacoordinated intermediate [Ir(SiNP)(cod)(CNtBu)][PF6] (2PF6) forms in the first place and that further reacts with CNtBu, affording the square planar intermediate [Ir(SiNP)(CNtBu)2][PF6] (3PF6) that finally undergoes the intramolecular oxidative addition of the SiCH2–H bond. The reactivity of 1PF6 was investigated. On one hand, the reaction of 1PF6 with N-chlorosuccinimide or N-bromosuccinimide provides the haloderivatives [IrX{κ3C,P,P′-(SiNP–H)}(CNtBu)2][PF6] (X = Cl, 4PF6; Br, 5PF6), and the reaction of 5PF6 with AgPF6 in the presence of acetonitrile affords the solvato species [Ir{κ3C,P,P′-(SiNP–H)}(CH3CN)(CNtBu)2]2+ (62+) isolated as the hexafluorophosphate salt. On the other hand, the reaction of 1PF6 with HBF4 gives the iridium(III) compound [IrH(CH2SiF2CH3)(HNP)2(CNtBu)2][BF4] (7BF4) as the result of the formal addition of hydrogen fluoride to the Si–N bonds of 1+ [HNP = HN(4-tolyl)PPh2]. A similar outcome was observed in the reaction of 1PF6 with CF3COOH rendering 7PO2F2. In this case the intermediate [IrH{κ2C,P-CH2SiMeFN(4-tolyl)PPh2}(HNP)(CNtBu)2]+ (8+) was observed and characterised in situ by NMR spectroscopy. DFT calculations suggests that the reaction goes through the sequential protonation of the nitrogen atom of the Si–N–P moiety followed by the formal addition of fluoride ion to silicon. Also, the crystal structures of SiNP, 1PF6, 4PF6 and 7BF4 have been determined by X-ray diffraction measurements.


 Please wait while we load your content...
Please wait while we load your content...