Hybrid computational–experimental data-driven design of self-assembling π-conjugated peptides†
Abstract
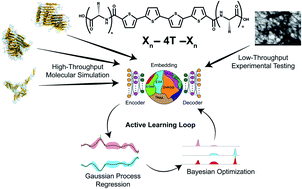
Biocompatible molecules with electronic functionality provide a promising substrate for biocompatible electronic devices and electronic interfacing with biological systems. Synthetic oligopeptides composed of an aromatic π-core flanked by oligopeptide wings are a class of molecules that can self-assemble in aqueous environments into supramolecular nanoaggregates with emergent optical and electronic activity. We present an integrated computational–experimental pipeline employing all-atom molecular dynamics simulations and experimental UV-visible spectroscopy within an active learning workflow using deep representational learning and multi-objective and multi-fidelity Bayesian optimization to design π-conjugated peptides programmed to self-assemble into elongated pseudo-1D nanoaggregates with a high degree of H-type co-facial stacking of the π-cores. We consider as our design space the 694 982 unique π-conjugated peptides comprising a quaterthiophene π-core flanked by symmetric oligopeptide wings up to five amino acids in length. After sampling only 1181 molecules (∼0.17% of the design space) by computation and 28 (∼0.004%) by experiment, we identify and experimentally validate a diversity of previously unknown high-performing molecules and extract interpretable design rules linking peptide sequence to emergent supramolecular structure and properties.


 Please wait while we load your content...
Please wait while we load your content...